CBSE Class 11 Nomenclature of Elements with Atomic Numbers > 100 Detail & Preparation Downloads
The nomenclature of elements with atomic numbers greater than 100 involves systematic naming conventions to identify and categorize these heavy elements. As discoveries in the realm of superheavy elements advance, a precise nomenclature ensures clarity and consistency in scientific communication. This introduction explores the evolving strategies employed in naming elements beyond atomic number 100, reflecting the ongoing exploration of the periodic table's upper reaches.
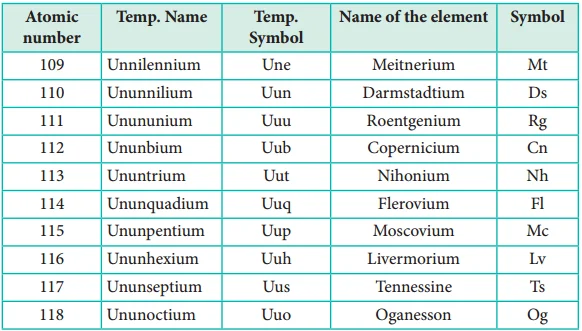
Nomenclature of Elements with Atomic Numbers > 100 - CBSE NCERT Download
How The Elements With The Atomic Number (Z) Above 100 Assigned Names?
The assignment of names to elements with atomic numbers (Z) above 100 involves a careful and systematic process governed by international guidelines established by the International Union of Pure and Applied Chemistry (IUPAC). Here's an overview of how elements with atomic numbers above 100 are assigned names:
-
Temporary Systematic Names:
- Before a new element is confirmed and officially named, it is assigned a temporary systematic name based on its atomic number. These names typically follow a systematic nomenclature convention.
- Examples include ununtrium (Uut) for element 113 and ununpentium (Uup) for element 115.
-
Confirmation of Discovery:
- Once the discovery of a new element is confirmed by multiple independent experiments, the researchers submit their findings to the IUPAC for review and verification.
-
Proposal for a Permanent Name:
- After confirmation, the discoverers propose a permanent name for the new element. The proposed name often honors a scientist, acknowledges a place, or reflects some key characteristic of the element.
-
IUPAC Evaluation:
- The proposed name undergoes evaluation by the IUPAC. The organization assesses the consistency of the name with existing naming conventions, ensuring it meets certain criteria and does not duplicate existing names.
-
Public Review:
- The proposed name is subjected to a public review period, during which the scientific community and the public can provide feedback. This step ensures a broader consensus and transparency in the naming process.
-
Official Approval:
- If the proposed name successfully passes the evaluation and public review, it is officially approved by the IUPAC. The new name is then added to the periodic table.
-
Incorporation into the Periodic Table:
- The approved name is incorporated into the periodic table, and the element is recognized globally by its official name. The element is typically represented by a symbol derived from its name.
-
Recognition of Contributors:
- The IUPAC may also consider recognizing the contributions of the scientists who discovered or synthesized the element by including their names or initials in the element's history.
The systematic and careful approach ensures consistency and accuracy in naming, reflecting the collaborative and international nature of scientific discovery and the importance of clear communication in the global scientific community.
Unveiling the Superheavy Elements:
The Quest for the Unknown:
As scientific exploration delves deeper into the cosmos, researchers uncover elements with atomic numbers exceeding 100. These superheavy elements, often created synthetically in laboratories, open new frontiers in understanding atomic structure and behavior.
Challenges in Nomenclature:
Naming these elements is no small feat. Traditional naming conventions, often inspired by mythology or honored scientists, continue, but the complexity of superheavy elements demands additional consideration. Standardizing their names ensures global coherence and facilitates communication in the scientific community.

CBSE Class 11th Downloadable Resources:
|
1. CBSE Class 11th Topic Wise Summary |
View Page / Download |
|
2. CBSE Class 11th NCERT Books |
View Page / Download |
|
3. CBSE Class 11th NCERT Solutions |
View Page / Download |
|
4. CBSE Class 11th Exemplar |
View Page / Download |
|
5. CBSE Class 11th Sample Papers |
View Page / Download |
|
6. CBSE Class 11th Question Bank |
View Page / Download |
|
7. CBSE Class 11th Topic Wise Revision Notes |
View Page / Download |
|
8. CBSE Class 11th Last Minutes Preparation Resources |
View Page / Download |
|
9. CBSE Class 11th Best Reference Books |
View Page / Download |
|
10. CBSE Class 11th Formula Booklet |
View Page / Download |
Being in CBSE class 11th and considering the board examinations you must be needing resources to excel in your examinations. At TestprepKart we take great pride in providing CBSE class 11th all study resources in downloadable form for you to keep you going.
Below is the list of all CBSE class 11th Downloads available on TestprepKart for both Indian and NRI students preparing for CBSE class 11th in UAE, Oman, Qatar, Kuwait & Bahrain.
SAMPLE PRACTICE QUESTIONS OF SIGNIFICANT FIGURES:
Q1. Why do elements with atomic numbers above 100 have temporary names?
Answer. Temporary names, often systematic, are assigned to elements during the initial stages of discovery to provide a standardized way of referring to them before their properties are fully studied and before the official naming process.
Q2. Who governs the official naming process for new elements?
Answer. The official naming process is governed by the International Union of Pure and Applied Chemistry (IUPAC), which establishes guidelines and evaluates proposed names for consistency and appropriateness.
Q3. How are permanent names proposed for new elements?
Answer. Discoverers propose permanent names based on factors like honoring scientists, geographical locations, or significant properties of the elements. The proposed names undergo rigorous evaluation by the IUPAC.
Q4. How are symbols derived for superheavy elements?
Answer. Symbols for elements are typically derived from their names. These symbols are often one- or two-letter abbreviations that are internationally recognized and added to the periodic table
Q5. Why is a systematic approach used for temporary names?
Answer. A systematic approach, based on atomic numbers, provides a standardized and logical way to refer to elements during the initial stages of discovery, facilitating communication within the scientific community.

| CBSE CLASS 11th Chemistry Chapters |
| Chapter1: SOME BASIC CONCEPTS OF CHEMISTRY |
| Chapter2: STRUCTURE OF ATOMS |
| Chapter3: CLASSIFICATION OF ELEMENTS AND PERIODICITY IN PROPERTIES |
| > Why do we Need to Classify Elements? |
| > Genesis of Periodic Classification |
| > Modern Periodic Law and the Present Form of the Periodic Table |
| > Electronic Configurations of Elements and the Periodic Table |
| > Electronic Configurations and Types of Elements: 82 s-, p-, d-, f- Blocks |
| > Periodic Trends in Properties of Elements |
| Chapter4: CHEMICAL BONDING AND MOLECULAR STRUCTURE |
| Chapter5: THERMODYNAMICS |
| Chapter6: EQUILIBRIUM |
| Chapter7: REDOX REACTIONS |
| Chapter8: ORGANIC CHEMISTRY - SOME BASIC PRINCIPLE AND TECHNIQUES |
| Chapter9: Hydrocarbons HYDROCARBONS |
| CBSE Class 11 Physics Chapters |
| Chapter1: UNITS AND MEASUREMENTS |
| Chapter2: MOTION IN A STRAIGHT LINE |
| Chapter3: MOTION IN A PLANE |
| Chapter4: LAWS OF MOTION |
| Chapter5: WORK, ENERGY AND POWER |
| Chapter6: SYSTEM OF PARTICLES AND ROTATIONAL MOTION |
| Chapter7: GRAVITATION |
| Chapter8: MECHANICAL PROPERTIES OF SOLIDS |
| Chapter9: MECHANICAL PROPERTIES OF FLUIDS |
| Chapter10: THERMAL PROPERTIES OF MATTER |
| Chapter12: KINETIC THEORY |
| Chapter13: OSCILLATIONS |
| Chapter14: WAVES |
| CBSE Class 11 Mathematics chapter |
| Chapter1: SETS |
| Chapter2: RELATIONS AND FUNCTIONS |
| Chapter3: TRIGONOMETRIC FUNCTIONS |
| Chapter4: COMPLEX NUMBER AND QUADRATIC EQUATIONS |
| Chapter5: LINEAR INEQUALITIES |
| Chapter6: PERMUTATIONS AND COMBINATIONS |
| Chapter7: BINOMIAL THEOREM |
| Chapter8: SEQUENCES AND SERIES |
| Chapter9: STRAIGHT LINES |
| Chapter10: CONIC SECTIONS |
| Chapter11: INTRODUCTION TO THREE-DIMENSIONAL GEOMETRY |
| Chapter12: LIMITS AND DERIVATIVES |
| Chapter13: STATISTICS |
| Chapter14: PROBABILITY |
| Class 8 Link soon |
| Class 9 Link soon |
| Class 10 Link soon |
| Class 12 Link soon |